8 (4) A, C 0.1 gm of organic compound was analysed by Kjeldahl's method. In analysis produced NH, absorbed in 30 ml N/5 H,SO. The remaining acid required 20 ml N/10 NaOH
Por um escritor misterioso
Last updated 15 junho 2024

Click here:point_up_2:to get an answer to your question :writing_hand:84 a c01 gm of organic compound was analysed bykjeldahls method in analysis produced nhabsorbed

An organic compound on analysis gave C=48gm,H=8gm and N =56gm. Volume of ..

The Titration in the Kjeldahl Method of Nitrogen Determination: Base or Acid as Titrant?

Methods for chemical analysis of soils - N.Z. Soil Bureau Scientific Reports - Manaaki Whenua Landcare Research Digital Library

Study Notes on Analytical Chemistry, EB 3247 - Analytical Chemistry - Nilai

The Titration in the Kjeldahl Method of Nitrogen Determination: Base or Acid as Titrant?

Co-operative Ecological Research Project (CERP), phase II: China - (mission). Project findings and recommendations

0.2 gm of an organic compound was analysed by kjeldahl's method the am

DPP (1 TO) Physical Chemistry

Study Notes on Analytical Chemistry, EB 3247 - Analytical Chemistry - Nilai
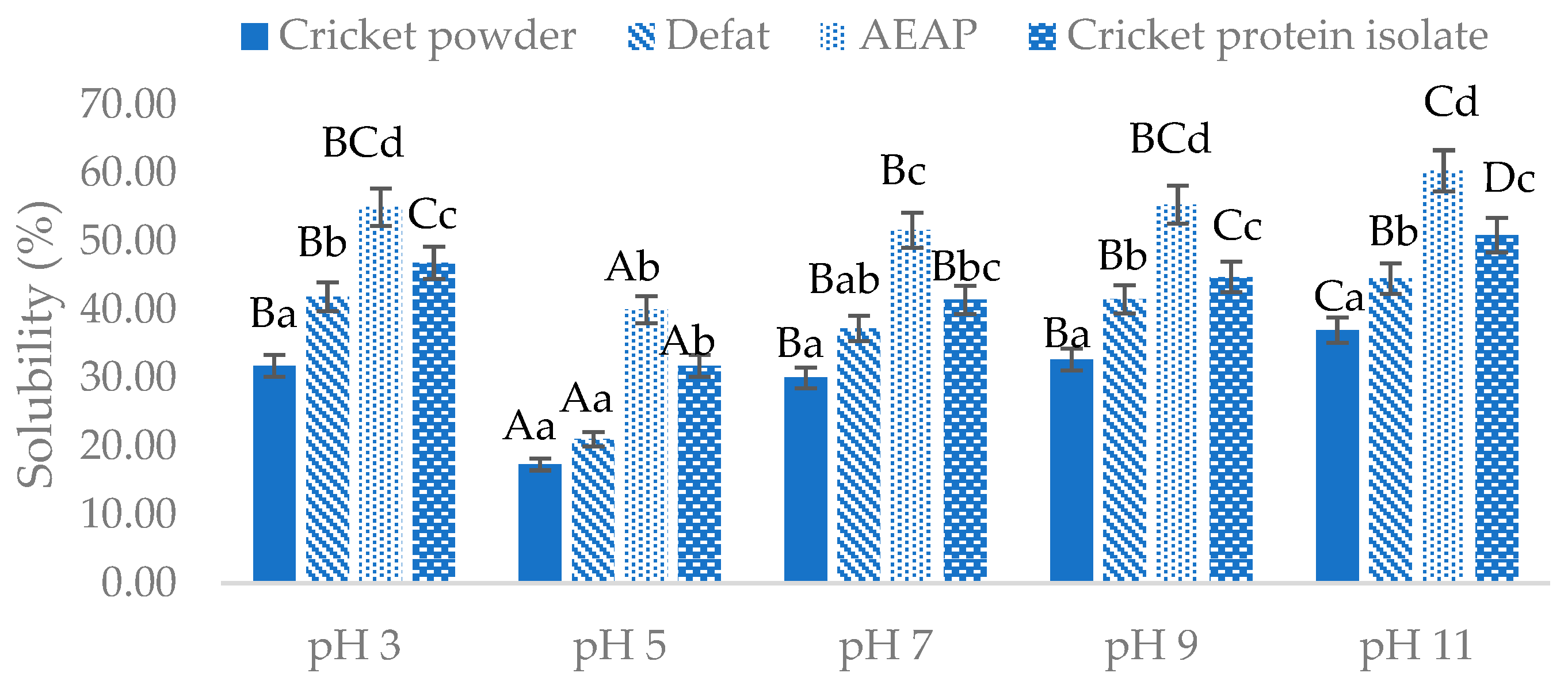
Foods, Free Full-Text

For the estimation of nitrogen, 1.4 g of an organic compound was digested by Kjeldahl method and the evolved ammonia was absorbed in 60 mL of M/10 sulphuric acid. The unreacted acid
Recomendado para você
-
 Proxies for Work-as-Done: 4. Work-as-Analysed – Humanistic Systems15 junho 2024
Proxies for Work-as-Done: 4. Work-as-Analysed – Humanistic Systems15 junho 2024 -
Analysed databases. Source. Web of Science Core Collection (2022).15 junho 2024
-
 Bloody, Bold and Resolute: Macbeth: Explained and Analysed (Paperback)15 junho 2024
Bloody, Bold and Resolute: Macbeth: Explained and Analysed (Paperback)15 junho 2024 -
 Mood board analysed15 junho 2024
Mood board analysed15 junho 2024 -
 Getting My Blood Analysed - How Healthy Am I? - Laura Try15 junho 2024
Getting My Blood Analysed - How Healthy Am I? - Laura Try15 junho 2024 -
 BUREAU OF ANALYSED SAMPLES LTD Certified Reference15 junho 2024
BUREAU OF ANALYSED SAMPLES LTD Certified Reference15 junho 2024 -
 Analysed Images15 junho 2024
Analysed Images15 junho 2024 -
 Affiliate SEO: We Analysed Who Ranks in 1,000 SERPs (2023)15 junho 2024
Affiliate SEO: We Analysed Who Ranks in 1,000 SERPs (2023)15 junho 2024 -
 Examples of analysed words and phrases15 junho 2024
Examples of analysed words and phrases15 junho 2024 -
 Videography: analysing video data as a 'focused' ethnographic and hermeneutical exercise - Hubert Knoblauch, Bernt Schnettler, 201215 junho 2024
Videography: analysing video data as a 'focused' ethnographic and hermeneutical exercise - Hubert Knoblauch, Bernt Schnettler, 201215 junho 2024
você pode gostar
-
Boruto Caderno Cartografia e Desenho Espiral Capa Dura 60 folhas15 junho 2024
-
 Mokele mbembe by cisiopurple on DeviantArt15 junho 2024
Mokele mbembe by cisiopurple on DeviantArt15 junho 2024 -
 Al Di Meola - Land of the Midnight Sun Album on HQ Vinyl15 junho 2024
Al Di Meola - Land of the Midnight Sun Album on HQ Vinyl15 junho 2024 -
 A criança aos 4 anos - Mãe-Me-Quer15 junho 2024
A criança aos 4 anos - Mãe-Me-Quer15 junho 2024 -
Todos os jogos da PG Soft #fortunetiger #fortuneox #pgsoft15 junho 2024
-
 Kamitachi ni Hirowareta Otoko Ch.43.5 Page 2 - Mangago15 junho 2024
Kamitachi ni Hirowareta Otoko Ch.43.5 Page 2 - Mangago15 junho 2024 -
 Crunchyroll Mod Apk v3.24.1 Premium freigeschaltet 202215 junho 2024
Crunchyroll Mod Apk v3.24.1 Premium freigeschaltet 202215 junho 2024 -
get you daniel caesar remix|Pesquisa do TikTok15 junho 2024
-
 Want to play Minecraft.Io? Play this game online for free on Poki15 junho 2024
Want to play Minecraft.Io? Play this game online for free on Poki15 junho 2024 -
 msm monster tier list, fight me : r/MySingingMonsters15 junho 2024
msm monster tier list, fight me : r/MySingingMonsters15 junho 2024


